In tracking skin care trends, it is evident that there are darker-skinned consumers who seek lighter skin tone, while many with lighter skin try their best to achieve a tanned, darker tone. The consistent desire, whatever the skin tone, is for evenness of color, which includes, in part, the elimination of age-related dark spots, hyperpigmentation and other skin pigmentation disorders. Brand owners are addressing this desire with products that lighten and brighten the skin—to even the skin color regardless of the tone. Following is an overview of the skin processes that impact tone/color, and ingredients and technologies that affect those processes and, therefore, skin tone.
The “M” Words— Think “Mechanisms”
Melanin, melanocytes, melanogenesis, melanosomes, melanocortins and MSH (melanocyte stimulating hormone)... Mother Nature created these and related mechanisms to provide humans with the skin tone that is best suited for their geography and living conditions. Briefly stated, MSH (produced in the pituitary gland) stimulates the production and release of melanin in skin and hair. An increase in MSH will cause a darkening of skin. In many redheaded and very light-skinned people (those who generally do not tan well), there are variations in their hormone receptors (melanocortins), causing them to not respond to MSH.
Melanin is a broad term for a group of natural pigments found in skin. Melanin is derived from the amino acid tyrosine and is produced in a specialized group of cells known as melanocytes through a process called melanogenesis. In the skin, melanogenesis occurs after exposure to UV radiation, causing the skin to visibly darken. Melanocytes insert granules of melanin into specialized cellular vesicles called melanosomes. These are then transferred into other skin cells of the human epidermis. The melanosomes in each recipient cell accumulate atop the cell nucleus, where they protect the nuclear DNA from mutations caused by the ionizing radiation of the sun’s ultraviolet rays.
There are three basic types of melanin: eumelanin, pheomelanin and neuromelanin. The most common type is eumelanin, and is produced in “black” and “brown” subtypes. Pheomelanin is a cysteine-containing red-brown polymer responsible for red hair and freckles. Neuromelanin, as its name implies, is a dark pigment that occurs in brain.1
New ingredients are constantly being developed to act on these mechanisms and alter the appearance of skin color and tone. Innovative delivery systems are required in topical formulations in order for these ingredients to reach melanocytes in skin. The efficacy of such ingredients can be significantly reduced or even lost if they penetrate too deeply or not at all into skin.
Brightening Activity, Ingredients and Formulation Basics
Let’s take a simplified look at which ingredients can or cannot function as skin brightening agents.
Tyrosine, an amino acid with a hydroxyphenyl chemical moiety, is the melanin precursor; this leads to L-DOPA (an amino acid with a dihydroxyaryl moiety), the ultimate precursor of melanin. The chemical basis of agonists and antagonists would suggest that any compound with a dihydroxyaryl moiety could function as a skin brightening and/or whitening agent. Even a hydroxyaryl (or a polyhydroxyaryl) compound can be a skin brightening agent (in view of tyrosine being a hydroxyaryl compound), provided it acts as an antagonist to tyrosine.
The most potent skin whitening agent, hydroquinone (to be discussed in more detail), has a dihydroxyaryl chemical backbone. Hydroxycinnamic acid3 also has a hydroxyaryl structure, and hundreds of flavones, isoflavones and flavonoids with this chemical feature have been shown to inhibit tyrosinase. Their use, however, has been limited due to their inherent bright color and oxidative instability.
Fundamentally, any antioxidant or anti-inflammatory agent can also be a skin brightening agent, as long as it can block the oxidation of L-DOPA into dopaquinone (i.e., inhibit tyrosinase). The efficacy of such ingredients can be manipulated by selecting appropriate delivery systems and use conditions. For example, hydroquinone is water-soluble, and the use of any formulation that contains hydroquinone prior to bathing or swimming can lead to significantly reduced skin brightening benefits. In this case, the nighttime use of such products can provide greater efficacy.
Formulation stability is another concern. Most hydroxy- and dihydroxyphenyl compounds tend to discolor in the presence of air and/or light. This is not all that unexpected, as most antioxidants tend to produce some discoloration after they have performed their intended function (if they do not already have some inherent color, due to their molecular structure). Usually, the greater the antioxidant or skin brightening efficacy, the faster this discoloration. This leads to ever-present formulation challenges with these ingredients. Phenolic ingredients including flavones, isoflavones and flavonoids are recommended to be formulated in an acidic pH range (preferably 5.5 to 6.5), as their degradation is faster in higher pH conditions.
New Skin Brightening Ingredients
In practical terms, the ongoing search for skin brightening ingredients and technologies has been successful via the pursuit of two basic biochemical mechanisms: either preventing melanin formation by blocking tyrosinase (tyrosinase inhibitors), or reducing existing melanin pigment (or melanin precursor intermediates with a quinone structure) to a lesser-colored species (melanin-reducing agents).
Hydroquinone can function via both mechanisms—it can block tyrosinase antagonistically by replacing tyrosine on the enzyme active site, or reduce melanin color by eliminating double-bond conjugation by donating two hydrogen atoms from its dihydroxyaryl backbone.
Ideally, a truly elegant new ingredient should function by the above two mechanisms without imparting any undesirable after-effects—such as skin irritation, which is caused by hydroquinone—irrespective of its highly desirable skin brightening efficacy. (In fact, the known skin irritancy of hydroquinone coupled with additional safety concerns has resulted in widespread and proposed bans on its non-physician managed topical use.) In general, the incorporation of more than one ingredient in a formulation is more beneficial for producing the desired skin brightening effect than a single ingredient on an equal weight basis, especially if such ingredients provide their biochemical function via different pathways.
Brightened Future for the Consumer
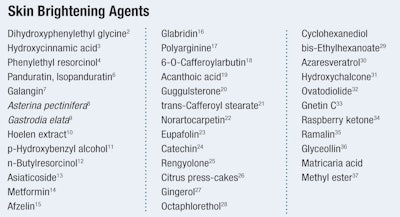
A select, non-comprehensive listing of both recently discovered and currently popular skin brightening agents is provided in Table 1. A number of these ingredients may not be commercially available at this time, making them an opportunity for ingredient manufacturers, formulators and marketers alike. An additional and substantial list of brightening ingredients is not discussed here, though well-explored in other articles.5 It is hoped that in this article, “what’s new and exciting” may lead to additional discoveries and their commercialization for skin care, to benefit marketers, formulators, ingredient suppliers and, ultimately, the consumer.
References
- en.wikipedia.org/wiki/Melanin
- Gupta, US Patent 7,777,073
- www.lipochemicals.com/products/lipobrite-hca-4
- products.symrise.com/life-essentials/actives/symwhiter-377
- S. Gupta, HAPPI, 90 (April 2001); Inside Cosmeceuticals (March 1, 2010); see also Gilbrow et al., Int J Cosmet Sci. 2011 Jun;33(3):210-21. doi: 10.1111/j.1468-2494.2010.00616.x. Epub 2011 Jan 25
- Yoon et al., Biol Pharm Bull. 2007 Nov; 30(11):2141-5; Lee et al., Phytother Res. 2010 Nov;24(11):1600-4. doi: 10.1002/ptr.3163. www.ncbi.nlm.nih.gov/pubmed/ 21031615; Phytother Res. 2010 Nov;24(11):1600-4. doi: 10.1002/ptr.3163
- Lu et al., J Enzyme Inhib Med Chem. 2007 Aug;22(4):433-8
- Jeong et al., Int J Mol Med. 2013 Jan;31(1):205-12. doi: 10.3892/ijmm.2012.1181. Epub 2012 Nov 14
- Kim et al., J Cosmet Sci. 2013 Mar-Apr;64(2):89-98
- Chang et al., Phytother Res. 2010 Sep;24(9):1359-64. doi: 10.1002/ptr.3123
- Liu et al., Biol Pharm Bull. 2007 Jun;30(6):1135-9
- Kim et al., Biol Pharm Bull. 2005 Dec;28(12):2216-9; Kolbe et al., J Eur Acad Dermatol Venereol. 2013 Jan;27 Suppl 1:19-23. doi: 10.1111/jdv.12051
- Kwon et al., Mol Med Rep. 2014 Apr 16. doi: 10.3892/mmr.2014.2159. [Epub ahead of print]
- Lehraiki et al., J Invest Dermatol. 2014 Apr 22. doi: 10.1038/jid.2014.202. [Epub ahead of print]
- Murata et al., Nat Prod Commun. 2014 Feb;9(2):185-8
- Hsieh et al., Br J Pharmacol. 2014 Feb 12. doi: 10.1111/bph.12626. [Epub ahead of print]; Simmler et al., Fitoterapia. 2013 Oct;90:160-84. doi: 10.1016/j.fitote.2013.07.003. Epub 2013 Jul 10
- Ookubo et al., Biomaterials. 2014 May;35(15):4508-16
- Xu et al., Nat Prod Res. 2014 Feb 5. [Epub ahead of print]. www.ncbi.nlm.nih.gov/pubmed/24498931
- Yoon et al., Nat Prod Commun. 2013 Oct;8(10):1359-62
- Koo et al., Int J Mol Med. 2012 Oct;30(4):974-8. doi: 10.3892/ijmm.2012.1057. Epub 2012 Jul 12
- Liang et al., Biol Pharm Bull. 2012;35(12):2198-203
- Ko et al., BMC Complement Altern Med. 2013 Dec 10;13:348. doi: 10.1186/1472-6882-13-348
- Ko et al., J Ethnopharmacol. 2014 Jan 10;151(1):386-93. doi: 10.1016/j.jep.2013.10.054. Epub 2013 Nov 7
- Khan et al., Postepy Dermatol Alergol. 2013 Aug;30(4):226-32. doi: 10.5114/pdia.2013.37032. Epub 2013 Aug 27
- Kim et al., Phytother Res. 2013 Nov 19. doi: 10.1002/ptr.5082. [Epub ahead of print]
- Kim et al., Asian Pac J Trop Biomed. 2013 Aug;3(8):617-22
- Huang et al., Biochem Biophys Res Commun. 2013 Aug 23;438(2):375-81. doi: 10.1016/j.bbrc.2013.07.079. Epub 2013 Jul 25
- Kim et al., Food Chem Toxicol. 2013 Sep;59:521-6. doi: 10.1016/j.fct.2013.06.031. Epub 2013 Jun 28
- Lim et al., Biol Pharm Bull. 2013;36(3):346-51. Epub 2012 Dec 17
- Franco et al., Molecules. 2012 Oct 9;17(10):11816-25. doi: 10.3390/molecules171011816
- Takahashi et al., J Oleo Sci. 2012;61(10):585-92
- Huang et al., Int J Mol Sci. 2012;13(5):6220-35. doi: 10.3390/ijms13056220. Epub 2012 May 21
- Yanagihara et al., Biol Pharm Bull. 2012;35(6):993-6
- Lin et al., Int J Mol Sci. 2011;12(8):4819-35. doi: 10.3390/ijms12084819. Epub 2011 Jul 28
- Paudel et al., Phytomedicine. 2011 Nov 15;18(14):1285-90. doi: 10.1016/j.phymed.2011.06.007. Epub 2011 Jul 29
- Lee et al.,BMB Rep. 2010 Jul;43(7):461-7. www.ncbi.nlm.nih.gov/pubmed/20663406
- Luo et al., Biol Pharm Bull. 2009 Jun;32(6):1091-4